
|
||||
| Home | Services | CDISC | Software | About us |
|
With classic EDC systems, the system has to be set up "by hand" for each new study that is started,
and the eCRFs need to be designed and created using information from the sponsor which is, at best, delivered in the form of Word or PDF documents.
Sponsors can however now design the whole study design using a user-friendly design tool that exports its results in CDISC ODM format.
This ODM file can be send to the EDC vendor, allowing the latter to set up the whole EDC system including the creation of eCRFs in a matter of minutes. The newest version of the CDISC ODM Standard (version 1.3) has a number of new features and enhancements directed to its use in EDC:
|
 Read our article in Applied Clinical Trials "CDISC Standards energize EDC" about the use of the CDISC ODM 1.3 standard in electronic data capture.
Read our article in Applied Clinical Trials "CDISC Standards energize EDC" about the use of the CDISC ODM 1.3 standard in electronic data capture.
As one of the main contributors to the development of the CDISC ODM 1.3 standard (especially the EDC-related new features), and our long-time involvement (>10 years) in the development of the CDISC ODM standard, XML4Pharma offers implementation services to EDC vendors, CROs and pharma companies:
Development of ODM Vendor Extensions
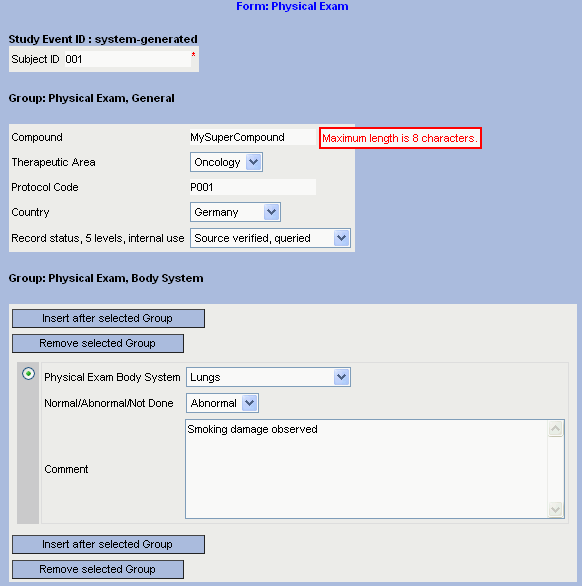
XML4Pharma has itself developed a new, fascinating technology for use in EDC. As we do not want to become an EDC Vendor ourselves, this technology is offered to EDC Vendors, CROs and pharma companies.
The technology is based on the automated transformation of a study setup in CDISC ODM format (v.1.2 or 1.3) into a set of eCRFs based on XForms technology.
XForms, an open standard itself, is the successor of web forms (HTML forms). Unlike web forms, XForms can be deployed on as well browsers, smartphones, tablets, etc., essentially on modern system, as the technology is platform-independent.There are however also many other advantages compared to HTML forms.
Our application server based technolgy accepts a CDISC ODM file and transforms it into a set of interactive eCRFs in XForms format (one for each Form definition in the CDISC ODM file). The eCRFs in XForms can then further styled and be deployed in an EDC system.
In our demo application, eCRFs are created on-the-fly from a CDISC ODM file, and can then be filled by the user. Upon submit, they are converted into CDISC ODM clinical data. For demo purposes, the obtained ODM is shown to the user. At the same time however, the ODM is also transformed into a PDF document (also shown to the user). In a real system, the latter can be implemented so that the PDF is stored in the investigators document management system, or is automatically printed on the investigators personal printer, or is simply shown to the investigator on the screen of his electronic device.
More about about automated creation of eCRFs directly from CDISC ODM files
View and try out some of the automatically generated eCRFs
